  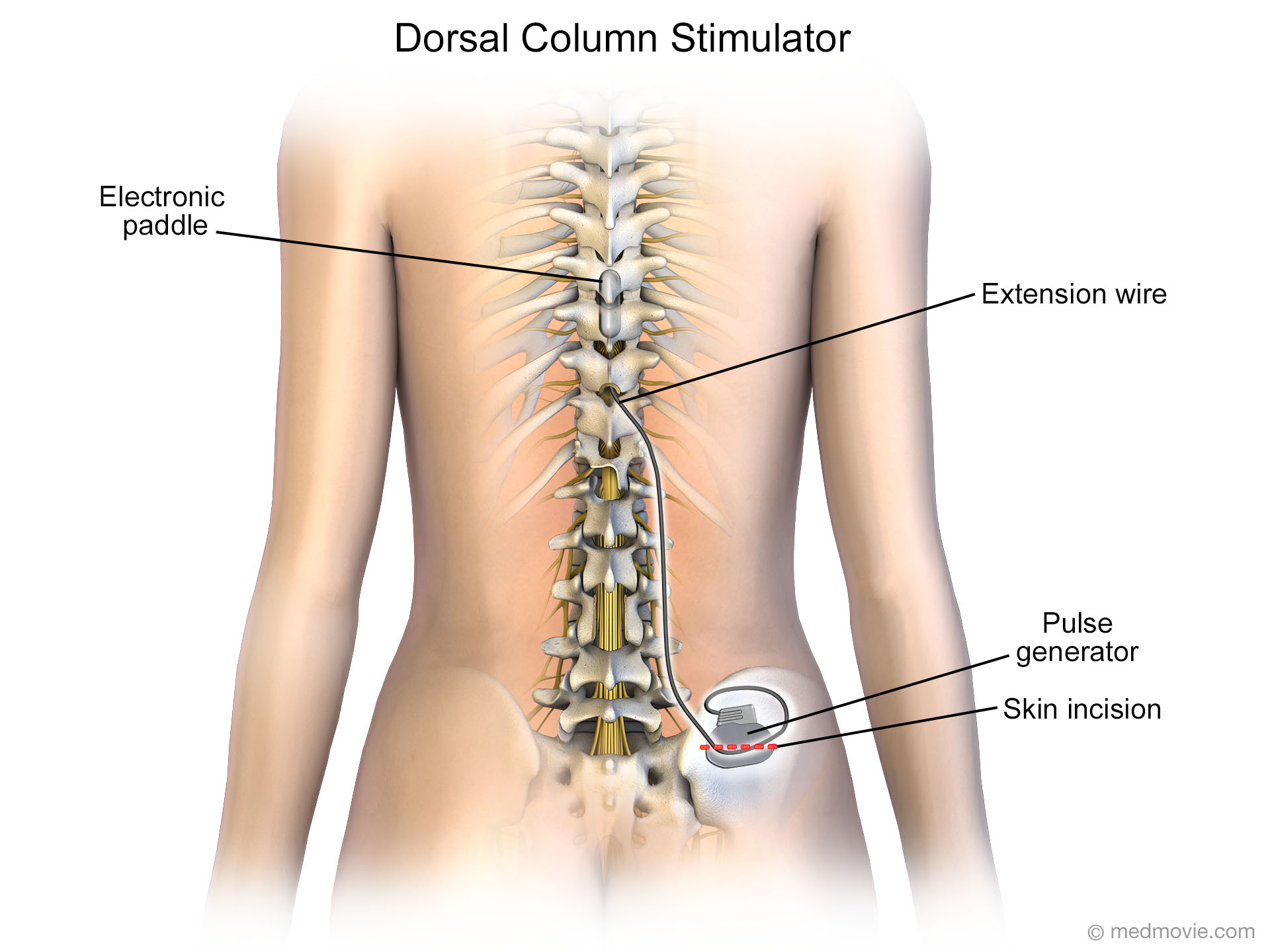
SCS may be contraindicated in people who have coagulation related disorders, or are on anticoagulant therapy. If pain control and increased activity was achieved, a permanent system, with leads and a pulse generator, is placed. Once a person has had a psychological evaluation and deemed an appropriate candidate for SCS, a temporary implant is placed, called a trial, to determine the best stimulation pattern, and the person is sent home for three to ten days with an external pulse generator. Īs of 2014 the FDA had approved SCS as a treatment for FBSS, chronic pain, complex regional pain syndrome, intractable angina, as well as visceral abdominal and perineal pain and pain in the extremities from nerve damage. The most common use of SCS is failed back surgery syndrome (FBSS) in the United States and peripheral ischemic pain in Europe. There are also spinal cord stimulators under research and development that could enable patients with spinal cord injury to walk again via epidural electrical stimulation (EES). SCS is a consideration for people who have a pain condition that has not responded to more conservative therapy. A spinal cord stimulator ( SCS) or dorsal column stimulator ( DCS) is a type of implantable neuromodulation device (sometimes called a "pain pacemaker") that is used to send electrical signals to select areas of the spinal cord (dorsal columns) for the treatment of certain pain conditions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
